Sponsored by Sica Media*
Friday July 18, 2025
OKYO Pharma (Nasdaq: OKYO)
👉OKYO is TODAY’S #1 ALERT 👈
Hey Folks,
Just a quick note before we head into the weekend…
As I hope you know, I’ve been on a roll with my “tactical trade” ideas.
Twelve of my last 16 ideas hit double-digit intraday gains on the days I alerted them, including yesterday’s 20% gainer.
Right now, I’m focused on an idea that climbed 30% the day I brought it to your attention last October, 16% the day I alerted you to it in January, and 14% when I alerted it last month.
💥Go ahead and pull up OKYO Pharma Limited (OKYO) on your trading platform.
Since my January 8 alert, the stock has climbed 143%. If you’ve been watching that one the whole time – congratulations!
As you can see, though, the action truly picked up as the stock rebounded with the rest of the market in early April…
Since its April 8 low, the stock is up 176%, and has been flirting with its 52-week high several times this month.
As I’ll cover more below, that’s been greatly fueled by late April/early May announcements of clinical breakthroughs for its lead candidate.
The stock has ripped as much as 26% higher over the last month, and with a big announcement out yesterday about an influx of non-dilutive funding, OKYO is primed for action — make sure it tops your watchlist today!
👉 OKYO is TODAY’S #1 ALERT 👈
In the meantime, here are some notes from my research about this interesting company…
To begin with, OKYO has an excellent website.
I’ve looked at tons of clinical-stage biopharma sites, and most are so full of jargon that it’s hard to see how any regular investor could understand them.
Maybe that’s their goal??
OKYO, on the other hand, does a great job of explaining their science, leadership, and investment opportunity.
The company’s main target is Dry Eye Disease (DED), which is expected to reach a $6.57 billion global market value by 2027.
DED results when the eyes don’t produce enough tears, causing poor lubrication that can lead to redness, grittiness, burning, blurring, and eye fatigue.
It’s a common condition, with approximately 38 million cases in the US and 700 million cases worldwide.
It affects over 35% of the population aged 50+, and two-thirds of DED patients are women.
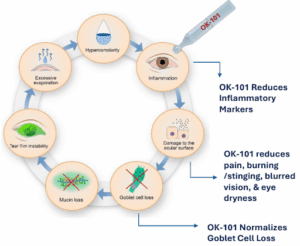
OKYO is rapidly advancing its lead candidate, urcosimod — a topical drug designed to control inflammation and pain in the eye.
Urcosimod was formerly known as OK-101 before it received its United States Adopted Name (USAN) in February.
The company began a Phase 2 trial for urcosimod as a treatment for DED in May 2023 and revealed ”extremely encouraging” topline data from the trial in January last year.
The trial found that urcosimod had a “highly favorable tolerability profile” and achieved statistical significance for multiple “sign” and “symptom” endpoints.
Specifically, the drug proved superior to the placebo for “total conjunctival staining” — a sign of DED — and for burning/stinging and blurred vision.
The company said that “To our knowledge, there are no FDA approved DED drugs that have been shown in clinical studies to improve conjunctival staining.”
OKYO now plans to advance urcosimod to a Phase 3 trial with the goal of developing a “highly differentiated dry eye product to help patients underserved by current treatments.”
In the course of the Phase 2 trial, researchers also found support for the potential of urcosimod to treat neuropathic corneal pain (NCP) — “a severe, chronic, and debilitating disease for which there are no approved commercial treatments currently available” [emphasis added]
As a result, OKYO is pursuing a parallel focus of urcosimod for that disease.
In February 2024, the FDA “cleared OK-101 as its first Investigational New Drug (IND) application for the treatment of NCP” and OKYO began a Phase II trial for that purpose last October.
The principal investigator of the trial is Dr. Pedram Hamrah of Tufts Medical Center — a renowned expert in NCP.
As OKYO CEO Gary Jacob, Ph.D. explained in an excellent October interview, the company has a “tremendous opportunity from a commercial standpoint” by pursuing a drug to treat a major unmet medical need. This “can lead to partnerships with big pharma and the kind of final merger acquisition outcome.”
On April 30, OKYO announced pivotal news of the early closure of its Phase 2 trial…
The company had planned to run the trial through the end of the year, but the early data was so promising, OKYO decided “to access the currently masked data and use it to plan its expanded development program.”
A number of trial patients even requested continued compassionate use of urcosimod.
“The decision to close the trial now significantly cuts the time to our requesting an end-of-phase 2 meeting with FDA to explore accelerated plans for the drug’s further clinical development,” explained OKYO CEO Gary Jacob.
For more on that, be sure to check out this April 30 interview with Mr. Jacob.
The next day, OKYO announced that the FDA granted urcosimod a Fast Track Designation for the treatment of NCP, which will allow more frequent meetings with the FDA as well as “eligibility for Accelerated Approval and Priority Review if relevant criteria are met, and a potential Rolling Review of the New Drug Application (NDA).”
And then just on Wednesday, OKYO revealed its “strong phase 2 clinical trial results for urcosimod to treat neuropathic corneal pain”
The company noted that “75% of patients treated with 0.05% urcosimod in this group achieved greater than 80% improvement in pain severity based on [Visual Analogue Scale] scores.”
Top-line data that strong for a treatment that targets an unmet medical need can be a total game-changer for a small company like OKYO.
Then yesterday, we learned OKYO had received “$1.9 million in non-dilutive funding to support its ongoing research and development programs.”
The company said the funding would help “advance key clinical milestones, expand R&D efforts, and build momentum behind urcosimod’s regulatory pathway.”
It’s worth noting that CEO Gary Jacob purchased 10,000 OKYO shares in January, a few months after the start of the NCP trial.
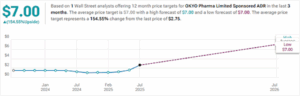
The sole analyst covering OKYO reiterated on July 16 a 12-month price target of $7.00 — a nearly 154% increase over the stock’s current price:
For your own research, I recommend starting with the company’s website and its June 2025 corporate presentation.
As always, be sure to approach your trading in a responsible manner. Trading is very risky, and nothing is ever guaranteed, so never trade with more than you can afford to lose.
Please read the full disclaimer at the bottom of this email as well, so you are aware of additional risks and considerations. Always have a well-thought-out game plan that takes your personal risk tolerance into consideration.
Bottom line: OKYO has been on a breakout since April 8 surging 176% to its current level and repeatedly flirting with its 52-week high.
With promising top-line data out Wednesday, news of new non-dilutive funding yesterday, I think OKYO could be primed for another ramp-up.
💥Make sure OKYO is leading your watchlist today!
To Your Success,
Jeff Bishop
*DISCLAIMER: This entity is owned by Sherwood Ventures LLC (SV). To more fully understand any SV subscription, website, application or other service, please review our full disclaimer located at https://bullseyealerts.com/disclaimer/
Just so you know, what you’re reading is curated content for which we have received a monetary fee (detailed below) to create and distribute. Let’s be clear that investing can be quite the roller coaster as stock prices can have wild swings up and down, so consider those crucial risks before you ever consider trading anything we discuss. Make sure you check out our full disclosure down below for the details on how we were paid, the risks, and why these results aren’t what you’d call “typical.”
Just a quick heads up about this ad you’re reading—as we’ve said, even though we like the company referenced above, and all the facts we discussed above are true to the best of our knowledge, we are running a business here. To distribute this information and help offset the costs of maintaining our large digital audience, in advance of writing the content above, we have been paid ten thousand dollars (cash) by bank transfer Sica Media for advertising OKYO Pharma Limited for a one day marketing program on July 18, 2025. Additionally, we have been paid twelve thousand five hundred dollars (cash) by bank transfer Sica Media for advertising OKYO Pharma Limited for a one day marketing program on June 20, 2025. Prior to this, we were paid twenty five thousand dollars (cash) by bank transfer by OKYO Pharma Limited for advertising for a one day marketing program on January 8, 2025. Previously, we also received paid twenty five thousand dollars (cash) by bank transfer by OKYO Pharma Limited for advertising for a one day marketing program on October 23, 2024. It might seem obvious, but while our client claims not to own any shares in OKYO Pharma Limited, whoever ultimately paid them most likely owns shares. You should assume they are looking to sell some or all of them at any time after we send out this information, which might negatively affect the stock price. We may also buy or sell shares in the company at some point in the future, although neither Sherwood Ventures nor its owners own any shares of the company at this time. Also, keep in mind that due to the sheer size of our audience, if even a small percentage of people decide they want to buy this stock, it could potentially boost interest enough to hike up those share prices and cause a temporary spike, and the opposite is possible as the marketing campaign ends, though that is not always the case.
Now, diving right into OKYO Pharma Limited might sound exciting. But remember, it’s like venturing into the wilderness—be aware that there’s exceptional risk involved in trading. This isn’t small potatoes we’re talking about; you could lose every dime you put in, so always carefully think about what you’re doing. That’s why they call this trading, after all. We’re shining a light on the good stuff about the company here, but it’s on you to do your homework, make your own calls, and determine a plan for your own trading, hopefully with the help of your professional 1nvestment advis0r.
Oh, that brings us to another crucial point—we’re not here to tell you (or even recommend) what you should do with your hard-earned money. We’re simply sharing our non-expert thoughts by highlighting some companies who are paying us and we like that could use some help telling their story to more people. We’re obviously biased in our writing. We’re not here to dig into anything that may be negative about the company; this is advertising, after all! Also, keep in mind that if we make some predictions about the future, these are technically known as “forward-L00king statements” under the securities acts, so take those with a grain of salt. As with all forecasts, they’re not set in stone, often wrong, and we certainly can’t know where the Company’s earnings, business, or share price will be tomorrow or a year from now.
Everything you read from us is all for your education, information, and possible entertainment. While we believe the info is reliable and accurate, we can’t wear a cape and guarantee it. Before you jump into anything, make sure to talk it over with a pro—someone you trust who’s licensed to give you real advice.
To be clear, Neither Sherwood Ventures nor its owners, employees, or independent contractors are registered as a securities broker-dealer, broker, 1nvest.ment advis0r (IA), or IA rep’s with the SEC, any state securities regulatory authority, or any self-regu1atory organization.
So, that’s the scoop! If you’re intrigued and want to learn more about the companies we talk about, hit up the SEC’s website to dig into their filings and see the full picture.